
Magnetic Nanoparticles Can Fix Broken Electric Wires
June 03, 2019 Inspired by armies of ants that link together to form living structures such as bridges and rafts, researchers have directed millions of nanoparticles to span the gap between two electrodes, forming an electrical wire (ACS Nano 2019, DOI: 10.1021/acsnano.9b02139). The technology could provide a new way to repair broken microcircuits and make tiny programmable microswitches, the researchers say.
Inspired by armies of ants that link together to form living structures such as bridges and rafts, researchers have directed millions of nanoparticles to span the gap between two electrodes, forming an electrical wire (ACS Nano 2019, DOI: 10.1021/acsnano.9b02139). The technology could provide a new way to repair broken microcircuits and make tiny programmable microswitches, the researchers say.
Roboticists dream of emulating the natural swarming behavior of insects and birds to make robots that work together on construction tasks or search-and-rescue missions. Scientists have also been engineering swarming particles that could have applications on the microscopic scale, such as tracking down tumors and delivering drugs.
Li Zhang, a mechanical and automation engineer at the Chinese University of Hong Kong, and his colleagues are designing microswarms for electronic applications. In 2018, they reported using oscillating magnetic fields to control millions of magnetic iron oxide nanoparticles in an ethanol suspension (Nat. Commun. 2018, DOI: 10.1038/s41467-018-05749-6). Changing the field’s strength relative to the fluid’s resistance made the particles line up into ribbon-like chains. By programming other parameters of the magnetic field, such as the oscillation frequency and angle, the researchers could elongate and shorten the ribbons, and make the nanoparticles split up and regroup. They could also make the swarms move through a maze.
Magnetic Bacteria Cause a Stir
May 19, 2019
Carrying out chemical reactions in Pickering emulsions avoids the large temperature and concentration gradients associated with traditional reaction systems. ‘But still, we need to find a way to stir the solution inside these micro-droplets. And this is the reason why we carried out this study to develop nano-sized stirrers,’ explains Weiguo Song from the Chinese Academy of Sciences.
For more information, check out https://www.chemistryworld.com/news/magnetic-bacteria-stir-reaction-in-droplet-microreactor/3008713.article.
Toyota Promises Cheaper Magnets for Electric Cars
May 19, 2019Japanese carmaker Toyota says it can cut the cost of rare earth element-containing magnets within the motors of electric vehicles by replacing up to half the neodymium in them with the more abundant, lower-cost rare earths lanthanum and cerium.
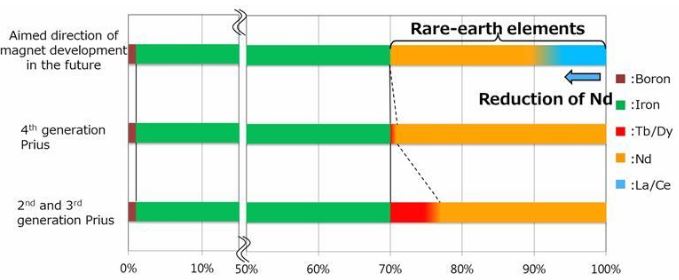
For more information, check here.
A Blood Test for Alzheimer's Disease Draws Near
March 28, 2019Anew approach with groundbreaking sensitivity is a major upgrade to a classic technique known as enzyme-linked immunosorbent assay (ELISA). The Massachusetts-based firm Quanterix has developed an automated version of ELISA that is 1,000 times as sensitive as older systems. Instead of tethering antibodies to the floor of wells in a microtiter plate, which usually holds multiple antibodies in each well, the company’s assay uses antibodies tethered to individual beads, which come to rest in individual wells on a grid. Each well is only 50 fL—about half a billionth the volume of a microtiter plate well—and holds only a single bead. By diluting plasma samples and adjusting the ratio of beads to protein molecules, researchers can ensure that each bead carries only one bound protein. An automated digital detection system measures the glow that results from a chemiluminescent reaction that occurs when the protein is present, allowing the assay to detect single protein molecules. These adjustments to dilution, reaction volumes, and standardization—coupled with antibodies that are highly specific to the peptide biomarkers—overcome problems including amyloid stickiness and ensure that newer assays outperform older attempts.
The Quanterix assay is 1,000-fold as sensitive as traditional enzyme-linked immunosorbent assays because it uses antibody-carrying magnetic beads that fit individually into femtoliter-volume wells. If a plasma protein of interest is present, the detection antibody linked to an enzyme binds to it. The enzyme goes on to cleave a chemiluminescent signaling molecule that can be detected in the well.
How Magnetic Nanoparticles Can Preserve the Historic Warship Mary Rose
February 20, 2019 On July 19, 1545, King Henry VIII of England watched in dismay as one of his greatest warships, the Mary Rose, sank during the Battle of the Solent in the strait between Britain’s south coast and the Isle of Wight, taking the lives of hundreds of sailors with it. The English fleet eventually won its battle against the French, but the Mary Rose remained on the seabed for centuries. Then, in 1982, the oak vessel was raised in a spectacular feat of underwater archaeology. The ship and its artifacts are now kept at the Mary Rose Museum in Portsmouth, England, but conservators face a battle of their own—preserving the ship’s oak timbers from decay. At the American Chemical Society national meeting in August, Serena Corr of the University of Sheffield explained how she is using nanotechnology to help save the Mary Rose. C&EN caught up with Corr to get the latest on the project.
On July 19, 1545, King Henry VIII of England watched in dismay as one of his greatest warships, the Mary Rose, sank during the Battle of the Solent in the strait between Britain’s south coast and the Isle of Wight, taking the lives of hundreds of sailors with it. The English fleet eventually won its battle against the French, but the Mary Rose remained on the seabed for centuries. Then, in 1982, the oak vessel was raised in a spectacular feat of underwater archaeology. The ship and its artifacts are now kept at the Mary Rose Museum in Portsmouth, England, but conservators face a battle of their own—preserving the ship’s oak timbers from decay. At the American Chemical Society national meeting in August, Serena Corr of the University of Sheffield explained how she is using nanotechnology to help save the Mary Rose. C&EN caught up with Corr to get the latest on the project.
Check out this article here.
Biomagnetic Particles Meeting 2019 in Telluride, Colorado, USA
February 14, 2019 Frontiers in Biomagnetic Nanoparticles that will be held August 5-7 in beautiful Telluride, Colorado, USA is now accepting abstracts. The program already includes 6 confirmed invited speakers, spanning Physics, Chemistry, Biology, Engineering and Medicine.
Frontiers in Biomagnetic Nanoparticles that will be held August 5-7 in beautiful Telluride, Colorado, USA is now accepting abstracts. The program already includes 6 confirmed invited speakers, spanning Physics, Chemistry, Biology, Engineering and Medicine.
There will be a number of travel scholarships to students and postdocs in need, by providing housing and registration for the meeting. To apply for the scholarships please send your application to abstracts@magneticnanoparticle.com. The application packet must be a single pdf file containing a) Statement of need (please keep less than 300 words), b) Letter of recommendation from research advisor, c) Abstract (please see abstract guidelines). Deadline for applying for these grants is March 1, 2019.
Check out further meeting information at https://www.magneticnanoparticle.com/.
Important Dates: Abstract Submissions: March 1, 2019; Registration Deadline: May 1, 2019
Magnetic Particle Imaging Conference in New York - March 17-19, 2019
February 14, 2019 Peter Caravan from the Athinoula A. Martinos Center for Biomedical Imaging, Department of Radiology, Massachusetts General Hospital, USA. With his keynote speech "MPI: The Future of Biomedical Molecular Imaging?” he shows how Magnetic Particle Imaging relates to existing modalities. Find the keynote abstract here: Sunday, March 17, 2019, 17:30 - 18:00 at Farkas Auditorium, NYU Langone Health.
Peter Caravan from the Athinoula A. Martinos Center for Biomedical Imaging, Department of Radiology, Massachusetts General Hospital, USA. With his keynote speech "MPI: The Future of Biomedical Molecular Imaging?” he shows how Magnetic Particle Imaging relates to existing modalities. Find the keynote abstract here: Sunday, March 17, 2019, 17:30 - 18:00 at Farkas Auditorium, NYU Langone Health.
Following topics of MPI will be presented at the conference: Medical, pre-clinical, and non-medical applications - Coil and field generator design - Data acquisition and signal pre-processing - Signal generation, amplification and filter design - Magnetic field simulation and system modeling - Magnetic particle spectroscopy (MPS) - Molecular imaging - Nanoparticle development - Particle physics and simulations - Reconstruction methods - Sequences, acquisition protocols and spatial coding - Scanner geometries and system design. More information can be found in the tentative program.
Novel Characterization Technique for Superparamagnetic Iron Oxide Nanoparticles
February 01, 2019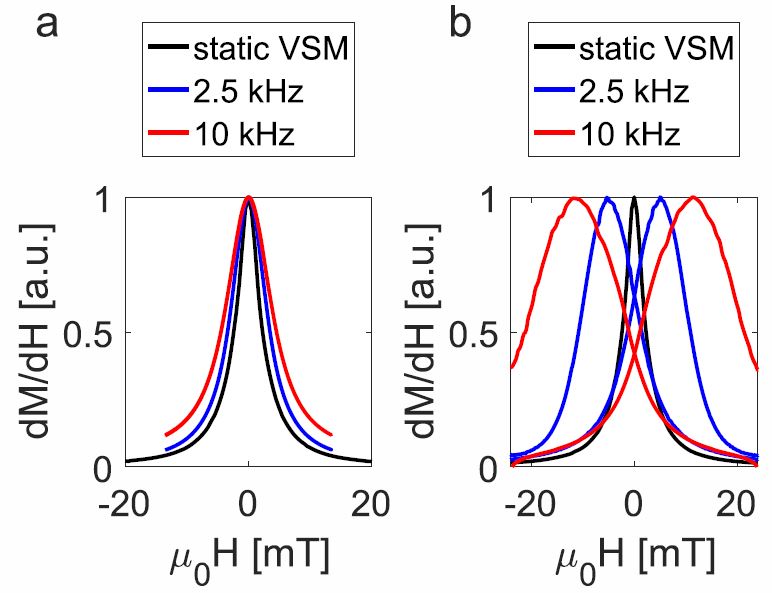
The magnetization curve was measured for two types of SPIONs: Resovist and SHP-25, using 3 techniques: Vibrating Sample Magnetometry (VSM), Magnetic Particle Spectroscopy (MPS), and our new SPaQ. Furthermore, AC susceptibility (ACS) measurements were performed as part of the evaluation of the 3 techniques. SPaQ and VSM results were found to be similar. Measurement results were nearly identical in both directions, indicating minor hysteresis. However, in MPS measurements, a clear hysteresis loop was observed. Furthermore, the ACS measurements showed a pronounced Brownian maximum, indicating an optimal response for an AC frequency below 10 kHz for both particle systems. Both the SPaQ and MPS were found to be superior to VSM since measurements are faster, can be performed at room temperature, and are particularly sensitive to particle dynamics. The main difference between the SPaQ and MPS lies in the excitation sequence. The SPaQ combines an alternating magnetic field that has a low amplitude with a gradual DC offset, whereas MPS uses only an alternating field that has a large amplitude.
Overall, both the SPaQ and MPS are highly suited to improve understanding SPION behavior! Check the paper out here.
For more information, check out our Archives.
September 2017

Search this site with the power of
