
Freshly printed magnets in 3D
August 15, 2020 Researchers at Empa, the Swiss Federal Laboratories for Materials Science and Technology near Zurich have developed an innovative method to precisely produce different alloy compositions in a single component, using a 3D laser printer. Until recently, 3D printing has presented unique challenges to metal processing, as temperatures nearly instantaneously exceed 2,500 degrees Celsius in the melting zone, which can alter the properties of alloys as some components evaporate. However, a team at Empa, led by Aryan Arabi-Hashemi and Christian Leinenbach, discovered that by varying the power and duration of the laser beam, they could modify the alloys with micrometer precision, thereby “producing new materials with completely new functionalities.” Prospective applications include manufacturing “smart” shape memory alloys and designing more efficient electric motors.
Researchers at Empa, the Swiss Federal Laboratories for Materials Science and Technology near Zurich have developed an innovative method to precisely produce different alloy compositions in a single component, using a 3D laser printer. Until recently, 3D printing has presented unique challenges to metal processing, as temperatures nearly instantaneously exceed 2,500 degrees Celsius in the melting zone, which can alter the properties of alloys as some components evaporate. However, a team at Empa, led by Aryan Arabi-Hashemi and Christian Leinenbach, discovered that by varying the power and duration of the laser beam, they could modify the alloys with micrometer precision, thereby “producing new materials with completely new functionalities.” Prospective applications include manufacturing “smart” shape memory alloys and designing more efficient electric motors.
The picture shows iron filings sticking to a mini chessboard with four millimeter edge length. The partially magnetic structure was produces from a single type of steel power at different temperatures. For more information, check it out here.
The World's Lightest Electromagnetic Shielding Material
August 14, 2020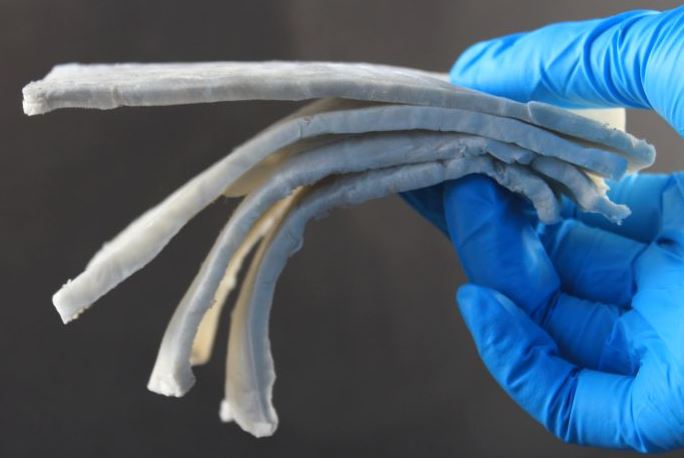
For more details, see here.
Microgripper Captures Single Cells with Magnetic Help
August 02, 2020.JPG)
For more information, and a movie, check it out here.
Special Issue Planned About Magnetic Cell Separation
June 02, 2020
The deadline for your manuscript submission will be November 30, 2020.
List of Cancelled Magnetic Conferences / Potential Jobs in Magnetism
May 01, 2020
http://magnetism.eu/news/144/38-news.htm
Also, while the Covid-19 situation is still dire, the curve seems flattened in many countries, and governments are carefully starting to reopen schools etc. This also means that more people are looking for jobs again.
In the magnetism area, EMA also updated their lists of available jobs. Please check it out too:
http://magnetism.eu/5-job-market.htm
And if you are an employer who is looking for a new employee, please advertise at EMA, or also send us an e-mail so we can advertise on magneticmicrosphere.com.
Virtual MPI Meetings Organized by Magnetic Insight
April 20, 2020
MPI is inviting researchers in the MPI field to participate in the Magnetic Particle Imaging Rising Stars 2020 Molecular Imaging eSymposia, taking place on April 21st and 22nd at 9 AM PT / 12 PM ET.
If you do some cool MPI research and would like to give a short 10 minute presentation, then please submit an abstract online at this address.
Abstract Deadline: April 3rd, 2020. For any questions, please contact Jeff Gaudet PhD.
Full-Color Magnetic Nanoparticles Based on Holmium-Doped Polymers
April 08, 2020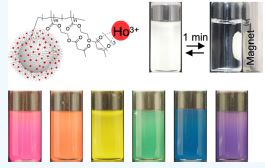
The bright and magnetically controllable colored nanoparticles presented herein may have a significant impact on practical substances and applications, such as ink and biomedical and device applications.
Check out the article here.
200th Anniversary of Hans Christian Oersted
April 07, 2020 On the occasion of the 200th anniversary of Hans Christian Ørsted's discovery of electromagnetism, a number of events were held during the year, including a series of anniversary lectures, which unfortunately had to be canceled due to the Coronavirus situation. Several of them have been video-recorded and can be viewed on YouTube.
On the occasion of the 200th anniversary of Hans Christian Ørsted's discovery of electromagnetism, a number of events were held during the year, including a series of anniversary lectures, which unfortunately had to be canceled due to the Coronavirus situation. Several of them have been video-recorded and can be viewed on YouTube.
As many of you know, I have a joint professorship at the Department of Pharmacy at the University of Copenhagen. Oersted was a pharmacist, and we can see him every day as a bronze bust (see to the right).
If you interested in his history, I recommend to watch now one of these lectures: HC Ørsted and the Pharmacy History, by Dr. Pharm. Poul R. Kruse, who worked at the Danish School of Pharmacy. With his usual legendary thoroughness and accuracy, Poul Kruse examines HC Ørsted's pharmaceutical background and decisive influence on pharmaceutical (and chemistry) education in Denmark in the 19th century, including the importance for the later development of the Danish pharmaceutical and chemical industry.
You can watch the lecture via this link: https://www.youtube.com/watch?v=oMIBqtR7f5Q
For more information, check out our Archives.
September 2017

Search this site with the power of
