
Special Issue Planned About Magnetic Cell Separation
June 02, 2020
The deadline for your manuscript submission will be November 30, 2020.
List of Cancelled Magnetic Conferences / Potential Jobs in Magnetism
May 01, 2020
http://magnetism.eu/news/144/38-news.htm
Also, while the Covid-19 situation is still dire, the curve seems flattened in many countries, and governments are carefully starting to reopen schools etc. This also means that more people are looking for jobs again.
In the magnetism area, EMA also updated their lists of available jobs. Please check it out too:
http://magnetism.eu/5-job-market.htm
And if you are an employer who is looking for a new employee, please advertise at EMA, or also send us an e-mail so we can advertise on magneticmicrosphere.com.
Virtual MPI Meetings Organized by Magnetic Insight
April 20, 2020
MPI is inviting researchers in the MPI field to participate in the Magnetic Particle Imaging Rising Stars 2020 Molecular Imaging eSymposia, taking place on April 21st and 22nd at 9 AM PT / 12 PM ET.
If you do some cool MPI research and would like to give a short 10 minute presentation, then please submit an abstract online at this address.
Abstract Deadline: April 3rd, 2020. For any questions, please contact Jeff Gaudet PhD.
Full-Color Magnetic Nanoparticles Based on Holmium-Doped Polymers
April 08, 2020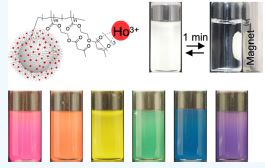
The bright and magnetically controllable colored nanoparticles presented herein may have a significant impact on practical substances and applications, such as ink and biomedical and device applications.
Check out the article here.
200th Anniversary of Hans Christian Oersted
April 07, 2020 On the occasion of the 200th anniversary of Hans Christian Ørsted's discovery of electromagnetism, a number of events were held during the year, including a series of anniversary lectures, which unfortunately had to be canceled due to the Coronavirus situation. Several of them have been video-recorded and can be viewed on YouTube.
On the occasion of the 200th anniversary of Hans Christian Ørsted's discovery of electromagnetism, a number of events were held during the year, including a series of anniversary lectures, which unfortunately had to be canceled due to the Coronavirus situation. Several of them have been video-recorded and can be viewed on YouTube.
As many of you know, I have a joint professorship at the Department of Pharmacy at the University of Copenhagen. Oersted was a pharmacist, and we can see him every day as a bronze bust (see to the right).
If you interested in his history, I recommend to watch now one of these lectures: HC Ørsted and the Pharmacy History, by Dr. Pharm. Poul R. Kruse, who worked at the Danish School of Pharmacy. With his usual legendary thoroughness and accuracy, Poul Kruse examines HC Ørsted's pharmaceutical background and decisive influence on pharmaceutical (and chemistry) education in Denmark in the 19th century, including the importance for the later development of the Danish pharmaceutical and chemical industry.
You can watch the lecture via this link: https://www.youtube.com/watch?v=oMIBqtR7f5Q
Magnetic Particle Spectroscopy for Detection of Influenza A Virus Subtype H1N1
April 06, 2020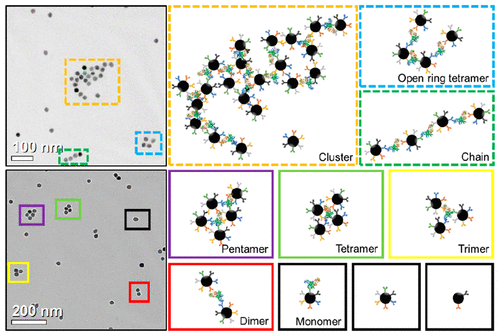 Magnetic nanoparticles (MNPs) with proper surface functionalization have been extensively applied as labels for magnetic immunoassays, carriers for controlled drug/gene delivery, tracers and contrasts for magnetic imaging, etc. Kai Wu and Jian-Ping Wang et al. introduced a new biosensing scheme based on magnetic particle spectroscopy (MPS) and the self-assembly of MNPs to quantitatively detect H1N1 nucleoprotein molecules. MPS monitors the harmonics of oscillating MNPs as a metric for the freedom of rotational process, thus indicating the bound states of MNPs. These harmonics can be readily collected from nanogram quantities of iron oxide nanoparticles within 10 s. The H1N1 nucleoprotein molecule hosts multiple different epitopes that forms binding sites for many IgG polyclonal antibodies. Anchoring IgG polyclonal antibodies onto MNPs triggers the cross-linking between MNPs and H1N1 nucleoprotein molecules, thereby forming MNP self-assemblies. Using MPS and the selfassembly of MNPs, they were able to detect as low as 44 nM (4.4 pmole) H1N1 nucleoprotein. In addition, the morphologies and the hydrodynamic sizes of the MNP self-assemblies are characterized to verify the MPS results. Different MNP self-assembly models such as classical cluster, open ring tetramer, and chain model as well as multimers (from dimer to pentamer) are proposed and form a new biosensing scheme for detecting ultralow concentrations of target biomolecules as a rapid, sensitive, and wash-free magnetic immunoassays. Check out more details in ACS Applied Materials & Interfaces.
Magnetic nanoparticles (MNPs) with proper surface functionalization have been extensively applied as labels for magnetic immunoassays, carriers for controlled drug/gene delivery, tracers and contrasts for magnetic imaging, etc. Kai Wu and Jian-Ping Wang et al. introduced a new biosensing scheme based on magnetic particle spectroscopy (MPS) and the self-assembly of MNPs to quantitatively detect H1N1 nucleoprotein molecules. MPS monitors the harmonics of oscillating MNPs as a metric for the freedom of rotational process, thus indicating the bound states of MNPs. These harmonics can be readily collected from nanogram quantities of iron oxide nanoparticles within 10 s. The H1N1 nucleoprotein molecule hosts multiple different epitopes that forms binding sites for many IgG polyclonal antibodies. Anchoring IgG polyclonal antibodies onto MNPs triggers the cross-linking between MNPs and H1N1 nucleoprotein molecules, thereby forming MNP self-assemblies. Using MPS and the selfassembly of MNPs, they were able to detect as low as 44 nM (4.4 pmole) H1N1 nucleoprotein. In addition, the morphologies and the hydrodynamic sizes of the MNP self-assemblies are characterized to verify the MPS results. Different MNP self-assembly models such as classical cluster, open ring tetramer, and chain model as well as multimers (from dimer to pentamer) are proposed and form a new biosensing scheme for detecting ultralow concentrations of target biomolecules as a rapid, sensitive, and wash-free magnetic immunoassays. Check out more details in ACS Applied Materials & Interfaces.
Astrophysicist Gets Magnets Stuck Up Nose While Inventing Coronavirus Device
March 30, 2020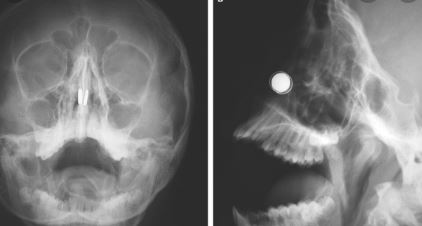
Dr Daniel Reardon, a research fellow at a Melbourne university, was building a necklace that sounds an alarm on facial contact, when the mishap occurred on Thursday night.
The 27 year-old astrophysicist, who studies pulsars and gravitational waves, said he was trying to liven up the boredom of self-isolation with the four powerful neodymium magnets.
“I have some electronic equipment but really no experience or expertise in building circuits or things,” he told Guardian Australia.
To read this article in its entirely, go on here.
3D Patterning of Cells in Magnetic Scaffolds for Tissue Engineering
February 26, 2020 Valentin Dediu et al. have realized a 3D magnetic patterning of two cell types in vitro inside an additive manufactured magnetic scaffold, as a conceptual precursor for the preparation of vascularised tissue. Nonhomogeneous magnetic gradients and loading magnetic configuration allowed for precise guiding of cells in a scaffold due to short range magnetic forces. The mathematical modelling confirmed the strong enhancement of the magnetic gradients and their particular geometrical distribution near the fibres, defining the preferential cell positioning on the micro-scale. Their method looks very promising in terms of assembling cellular constructs in a way that mimics physiological arrangements in vivo.
Valentin Dediu et al. have realized a 3D magnetic patterning of two cell types in vitro inside an additive manufactured magnetic scaffold, as a conceptual precursor for the preparation of vascularised tissue. Nonhomogeneous magnetic gradients and loading magnetic configuration allowed for precise guiding of cells in a scaffold due to short range magnetic forces. The mathematical modelling confirmed the strong enhancement of the magnetic gradients and their particular geometrical distribution near the fibres, defining the preferential cell positioning on the micro-scale. Their method looks very promising in terms of assembling cellular constructs in a way that mimics physiological arrangements in vivo.
Check the paper which was published in Scientific Reports out here.
For more information, check out our Archives.
September 2017

Search this site with the power of
