
Record-Breaking Molecular Magnet
March 13, 2022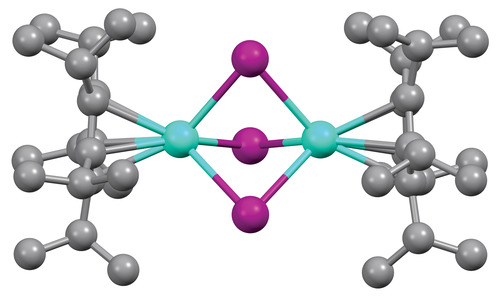 By coupling a pair of lanthanide ions within the same compound, researchers have created what they believe are the most magnetic molecules ever made.
By coupling a pair of lanthanide ions within the same compound, researchers have created what they believe are the most magnetic molecules ever made.
“By all the traditional metrics of single-molecule magnets, they’re the best,” Nicholas Chilton says of the new molecules. Chilton, who’s based at the University of Manchester, collaborated on the work with Jeffrey Long at the University of California, Berkeley, and Benjamin Harvey at the US Naval Air Warfare Center Weapons Division. Although the molecules’ magnetism reveals itself only at low temperatures, Chilton hopes that these dilanthanide complexes might pave the way for new types of powerful yet lightweight permanent magnets.
Lanthanides such as neodymium and samarium partner with transition metals in the strongest rare earth magnets, which are used in some electric vehicle motors and wind turbines. In rare earth magnets, metal-metal bonds help align unpaired electrons in the lanthanides and their transition-metal partners, boosting the overall magnetism. Coupling two lanthanides in this way should lead to even greater magnetism, but it has proved difficult to forge bonds between them.
Although lanthanides have previously bonded together inside fullerenes, Chilton says, “as far as I’m aware, these are the first conventional molecular lanthanide-lanthanide bonds.” The new complexes contain a pair of lanthanide ions—terbium or dysprosium, for example—bridged by three iodide anions and capped by bulky aromatic ligands. A single, shared electron sits in a bonding orbital between the two ions, and this helps align all the unpaired electrons on both ions.
For more details, check the article here: https://www.science.org/doi/10.1126/science.abl5470.
Magnetic Guiding with Permanent Magnets
October 09, 2021
What's included in this paper: The equations of magnetic fields and forces as well as velocities are derived in detail and physical limits are discussed. The special hydrodynamics of nanoparticle dispersions under these circumstances is reviewed and related to technical constraints.The possibility of 3D guiding and magnetic imaging techniques are discussed. Finally, the first results in guiding macroscopic objects, superparamagnetic nanoparticles, and cells with incorporated nanoparticles are presented. The constructed magnet systems allow for orientation, movement, and acceleration of magnetic objects and, in principle, can be scaled up to human size.
Check out this interesting article here.
European Inventor Price 2021 Goes to Two of Our Own
June 18, 2021 The European Patent Office honours Robert Grass and Wendelin Stark, professors of chemical engineering for their research in DNA encapsulation. Their invention enables the storage of digital data for thousands of years and product tracing throughout supply chains.
The European Patent Office honours Robert Grass and Wendelin Stark, professors of chemical engineering for their research in DNA encapsulation. Their invention enables the storage of digital data for thousands of years and product tracing throughout supply chains.
"DNA has a really high storage capacity," says Robert Grass. "A whole library can be stored in a really tiny amount of DNA for a millennia." Grass, professor and chemical engineer and Professor Wendelin Stark, who heads up the Functional Materials Engineering Lab at ETH Zurich are pioneers of DNA encapsulation. Masterfully combining nature with patented technology, their invention provides a novel method for synthesizing digital data into artificial DNA. The process even includes coding for error-correction contributed by fellow ETH Zurich engineer and data scientist, at the time, Reinhard Heckel. The team also achieved error-free data recovery, even after environmental exposure tests that simulate the equivalent of thousands of years of storage.
Developing nanometer-sized glass beads with diameters 10,000 times thinner than a sheet of paper, the beads protect the DNA from the corrosive forces of nature in much the same way that amber protects fossilized insects. Their invention enables digital DNA storage for thousands of years, meeting challenging data storage needs as technology accelerates at an exponential rate.
Eager to demonstrate the vast potential applications of their novel technology, Grass and Stark partnered up with English band, Massive Attack to commemorate the 20th anniversary of their Mezzanine album by encoding it in DNA. The album’s DNA was also incorporated into spray paint used to create an artwork with literally thousands of DNA copies of the album embedded in the paint on canvas work. They also encoded in DNA the premiere season of the Netflix television series, Biohackers demonstrating the versatility of the technology.
The glass-encapsulated DNA storage method that the professors and their research team have been developing since 2012 has also been commercialised by the former doctoral students Michela Puddu and Gediminas Mikutis with the ETH spin-off, Haelixa AG which they co-founded in 2016. At Haelixa, DNA is being used to label products such as precious stones, gold, and even organic cotton, to ensure that product origins or working conditions - are traceable throughout the supply chain. Grass and Stark have also co-founded several other companies including ETH spin-offs TurboBeads LLC and hemotune AG.
Microvehicles to Navigate Blood Vessels Upstream
April 17, 2021
Check out the details here, with movie: https://www.nature.com/articles/s42256-020-00275-x
Ultrafast Terahertz Magnetometry Could Enable Heat-Free Magnetism Control
March 30, 2021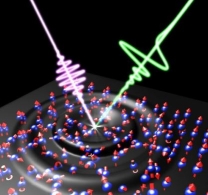
The work could pave the way for spintronic research and technologies based on a purely acoustic and, where possible, heat-free, ultrafast control of magnetism.
This principle states that a change in the magnetization of a material must result in an emission of electromagnetic radiation that will contain complete information on this change. If the magnetization in a material changes on a picosecond timescale, the emitted radiation will belong to the THz frequency range. This radiation, known as magnetic dipole emission, is weak and can be easily obscured by light emission that has originated elsewhere.
For more information, go here.
Special JMMM Issue Available - Scientific and Clinical Applications of Magnetic Carriers
March 21, 2021 While the 2020 meeting could not take place, you still had the opportunity of submitting a magnetic particle related article for a special JMMM issue, as we have done after all recent meetings. Twenty-five original articles are now available at this link. Get a coffee and some cake, and look through these interesting articles, they are well worth it!
While the 2020 meeting could not take place, you still had the opportunity of submitting a magnetic particle related article for a special JMMM issue, as we have done after all recent meetings. Twenty-five original articles are now available at this link. Get a coffee and some cake, and look through these interesting articles, they are well worth it!
And thank you Silvio Dutz and Maciej Zborowski for being the editor for this special issue.
www.sciencedirect.com/journal/journal-of-magnetism-and-magnetic-materials/special-issue/10L1Q3Q882M
European School on Magnetism (ESM) Open for Enrollment
February 28, 2021The next session of the European School on Magnetism (ESM) is scheduled to take place in Cluj-Napoca, Romania, 6-17 Sep 2021. It will be organized in a mixed format, on-site and on-line. This shall allow all to participate whatever their wish or ability to travel at the time, and also beyond the onsite hosting capabilities for the waiting list. Nevertheless, we feel ready to turn it fully on-line if sanitary conditions requested it.
As with previous sessions of ESM, the 2021 School aims at providing a thorough insight into magnetism through a broad series of fundamental lectures, and to address a specific topic of current interest in more detail. The topic chosen for the 2021 School is: “From fundamental properties of matter to magnetic materials and applications”. We intend to cover modern aspects of bulk and layered materials, both about their fundamentals and their applications.
The School is addressed mainly at PhD students and post-docs, both experimentalists and theoreticians. It will consist of approximately 40 hours of lectures, student projects conducted in small groups, practicals and tutorials, open question sessions (5-10h), and free access to a library on magnetism. Attendees will be able to present their own work during poster sessions. The detailed program is available from the web site: http://magnetism.eu/school/2021/program.
Request for participation to the school opens today. Potential attendees are expected to provide, along with their application form, a motivation letter and, for PhD students and post-docs, a recommendation letter from their supervisor. For more details, check out this flyer here.
Embracing defects and disorder in magnetic nanoparticles: the emergence of defect-engineering as a new approach to optimize particle properties
February 19, 2021.JPG)
The authors Aidin Lak, Sabrina Disch & Philipp Bender emphasize the beneficial impact of defects and disorder on intracellular magnetic hyperthermia performance of magnetic nanoparticles for drug delivery and cancer therapy. This report illustrates that defect-engineering in iron oxide nanoparticles emerges to become an alternative approach to tailor their magnetic properties for biomedicine, as it is already common practice in established systems such as semiconductors and emerging fields including perovskite solar cells. Additionally, the authors offer their perspectives and thoughts on how to deliberately induce defects in iron oxide nanoparticles and their potential implications for magnetic tracers to monitor cell therapy and immunotherapy by magnetic particle imaging.
This forward-looking progress report can be found on Lak, A., Disch, S., Bender, P., Embracing Defects and Disorder in Magnetic Nanoparticles. Adv. Sci. 2021, 2002682. https://doi.org/10.1002/advs.202002682
For more information, check out our Archives.
September 2017

Search this site with the power of
