Research
Dr. Hafeli's research aims to improve drug targeting in cancer therapy. To do this, he is developing radioactive pharmaceuticals (= radiopharmaceuticals), determining their microdosimetry, and then delivering them to different organs and tumours. A particularly fascinating approach to deliver pharmaceuticals that his lab is investigating is magnetic drug targeting. For this purpose, magnetic nanoparticles and microspheres are made that can be physically targeted to cancer cells using magnetic fields.Radiopharmaceuticals
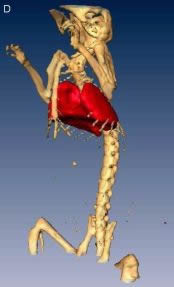 Radioactive drugs can be used for many therapeutic purposes such as cancer treatment or scar prevention. Radiopharmaceuticals, the general name for radiolabeled diagnostic and therapeutic agents, can take many different shapes including particles sized from tens of nanometers (= nanospheres) up to about 100 micrometers (= microspheres), viscous solutions and micellar/liposomal suspensions, sheets, and even metal implants such as stents (metal or plastic coils) or foils. Our lab is interested in preparing radioactively labelled drug delivery vehicles and using them to kill tumours and prevent their reoccurrence. The image to the left shows a radiopharmaceutical that we recently prepared and tested as a lung perfusion imaging agent. In a healthy animal, it perfectly outlines the lungs, without uptake in any other organ.
Radioactive drugs can be used for many therapeutic purposes such as cancer treatment or scar prevention. Radiopharmaceuticals, the general name for radiolabeled diagnostic and therapeutic agents, can take many different shapes including particles sized from tens of nanometers (= nanospheres) up to about 100 micrometers (= microspheres), viscous solutions and micellar/liposomal suspensions, sheets, and even metal implants such as stents (metal or plastic coils) or foils. Our lab is interested in preparing radioactively labelled drug delivery vehicles and using them to kill tumours and prevent their reoccurrence. The image to the left shows a radiopharmaceutical that we recently prepared and tested as a lung perfusion imaging agent. In a healthy animal, it perfectly outlines the lungs, without uptake in any other organ.
The main radioactive isotopes we are currently investigating are the beta-emitters rhenium-188 (Re-188), yttrium-90 (Y-90), and their diagnostic counterparts technetium-99m (Tc-99m), indium-111 (In-111) and gallium-67 (Ga-67). Some PET isotope are also included, such as fluorine-18 (F-18), gallium-68 (Ga-68), and zirconium-89 (Zr-89). We also use other isotopes such as iodine-125 (I-125), iodine-123 (I-123), palladium-103 (Pd-103) and iridium-192 (Ir-192).
Imaging With Our Own MicroPET/SPECT/CT Imager
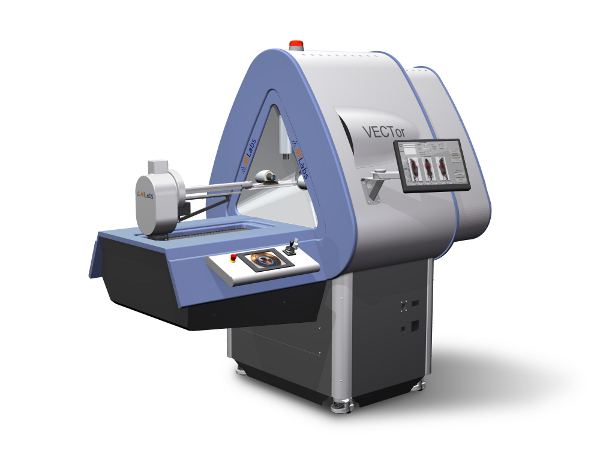 Together with Vesna Sossi (Physics) and Paul Schaffer (TRIUMF), we were successful in obtaining a Leaders Opportunity Fund grant titled "Hybrid microPET/SPECT and CT preclinical imaging". This allowed us to purchase a MILABS VECTor/CT instrument to UBC which allows now for simultaneous PET and SPECT imaging at resolutions not previously achievable. To see its capabilities, check out our imaging website. The instrument has been installed in July 2012 at the new Centre for Comparative Medicine (CCM) and is connected to TRIUMF with a direct underground hydraulic tube line from their radioisotope production facility. This pressurized tube allows the delivery of vials with freshly prepared radioactive isotopes directly from TRIUMF to the imaging room in the CCM facility. Many of the radioisotopes used in imaging are short lived (e.g., C-11 has a half-life of 20 min, N-13 of 10 min) and are, thus, only useful with ultrarapid transport and chemistry. The new scanner will be used to develop novel radiopharmaceuticals, to investigate diseases, to carry out brain research, and to enhance imaging physics and processing.
Together with Vesna Sossi (Physics) and Paul Schaffer (TRIUMF), we were successful in obtaining a Leaders Opportunity Fund grant titled "Hybrid microPET/SPECT and CT preclinical imaging". This allowed us to purchase a MILABS VECTor/CT instrument to UBC which allows now for simultaneous PET and SPECT imaging at resolutions not previously achievable. To see its capabilities, check out our imaging website. The instrument has been installed in July 2012 at the new Centre for Comparative Medicine (CCM) and is connected to TRIUMF with a direct underground hydraulic tube line from their radioisotope production facility. This pressurized tube allows the delivery of vials with freshly prepared radioactive isotopes directly from TRIUMF to the imaging room in the CCM facility. Many of the radioisotopes used in imaging are short lived (e.g., C-11 has a half-life of 20 min, N-13 of 10 min) and are, thus, only useful with ultrarapid transport and chemistry. The new scanner will be used to develop novel radiopharmaceuticals, to investigate diseases, to carry out brain research, and to enhance imaging physics and processing.
Magnetic Microspheres
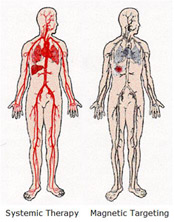 The main problem of cancer therapy is not the lack of efficient drugs, but that these drugs are very difficult to concentrate in the tumour tissue without leading to toxic effects on neighbouring organs and tissues. One method to accomplish this is by magnetic drug delivery with particulate carriers, a very efficient method of delivering a drug to a localized disease site. The figure to the left highlights the concept of magnetic targeting by comparing it with systemic drug delivery. In magnetic targeting, a drug or therapeutic radioisotope is bound to a magnetic compound, injected into a patient’s blood stream, and then stopped with a powerful magnetic field in the target area. Depending on the type of drug, it is then slowly released from the magnetic carriers (e.g., release of chemotherapeutic drugs from magnetic microspheres) or confers a local effect (e.g., irradiation from radioactive microspheres; hyperthermia with magnetic nanoparticles). Small amounts of drug targeted magnetically to localized disease sites can thus possibly replace large amounts of freely circulating drug and reach effective and up to several-fold increased localized drug levels.
The main problem of cancer therapy is not the lack of efficient drugs, but that these drugs are very difficult to concentrate in the tumour tissue without leading to toxic effects on neighbouring organs and tissues. One method to accomplish this is by magnetic drug delivery with particulate carriers, a very efficient method of delivering a drug to a localized disease site. The figure to the left highlights the concept of magnetic targeting by comparing it with systemic drug delivery. In magnetic targeting, a drug or therapeutic radioisotope is bound to a magnetic compound, injected into a patient’s blood stream, and then stopped with a powerful magnetic field in the target area. Depending on the type of drug, it is then slowly released from the magnetic carriers (e.g., release of chemotherapeutic drugs from magnetic microspheres) or confers a local effect (e.g., irradiation from radioactive microspheres; hyperthermia with magnetic nanoparticles). Small amounts of drug targeted magnetically to localized disease sites can thus possibly replace large amounts of freely circulating drug and reach effective and up to several-fold increased localized drug levels.
 In our lab, we are currently investigating new ways of preparing uniform magnetic microspheres with a microchip based flow focusing method. In this method, the polymer is dissolved and pumped through an orifice where the liquid stream breaks up into monosized droplets. The solvent is then extracted into the surrounding water phase, so that each droplet turns into a microsphere made from biodegradable materials and appropriate for human use. We are also interested in evaluating the toxicity of the whole microspheres and the small magnetic nanoparticles which give them their magnetic properties. These investigations are based on cell survival experiments and confocal microscopy investigations over time in cell cultures.
In our lab, we are currently investigating new ways of preparing uniform magnetic microspheres with a microchip based flow focusing method. In this method, the polymer is dissolved and pumped through an orifice where the liquid stream breaks up into monosized droplets. The solvent is then extracted into the surrounding water phase, so that each droplet turns into a microsphere made from biodegradable materials and appropriate for human use. We are also interested in evaluating the toxicity of the whole microspheres and the small magnetic nanoparticles which give them their magnetic properties. These investigations are based on cell survival experiments and confocal microscopy investigations over time in cell cultures.


