
Next Magnetic Carrier Meeting 2024 Will Take Place in Barcelona, Spain
December 03, 2022The next and 14th International Conference on the Scientific and Clinical Applications of Magnetic Carriers will take place in Barcelona, Spain. We will start on Monday evening with a reception, do our traditional boat trip on Thursday evening, and end the meeting on Friday. In-between, as always, we will dive deep into all the magic and new results and findings related to magnetic particles of all kinds. We are looking forward to have you all join us once more in this wonderful field!
For a bit more information (and much more to come later), check out this link.
2024 School of Magnetism in York, U.K.
February 12, 2024 ESM 2024 will be held from 27 August to 6 September 2024, in mixed format, both onsite in York, UK, and online. The topic is Magnetism for energy-efficient devices. Activities consist of lectures (40h), question sessions, practicals and tutorials, career and industry perspectives, access to a Library on Magnetism.
ESM 2024 will be held from 27 August to 6 September 2024, in mixed format, both onsite in York, UK, and online. The topic is Magnetism for energy-efficient devices. Activities consist of lectures (40h), question sessions, practicals and tutorials, career and industry perspectives, access to a Library on Magnetism.
The topic for the 2024 European School on Magnetism is Magnetism for energy-efficient devices. The School is chaired by Stuart Cavill (University of York).
Participation is offered in mixed format, either onsite in York, with a maximum of 100 participants, or online with most of the onsite activities available. We are commited to provide a similarly-high experience to both onsite and online participants, based on our experience of hybrid school built up since 2021. We intend to set up local hubs in Europe, in which online participants may gather to attend the school. This intends to enhance their online experience through collaborative work and emulation, and the mentoring of local experts in the field of magnetism. Hubs will be organized when the outcome of the selection process is over.
Like previous editions of ESM, the 2024 School aims at providing a thorough understanding of magnetism based on a broad series of fundamental lectures, while offering the latest insights into up-to-date aspects of magnetism with lectures focusing on a special topic. The objective of the 2024 school is to cover a wide range of fundamental to functional phenomena, to serve as a critical evaluation of the use of magnetism and magnetic materials to serve in energy-efficient devices, especially to address societal challenges. The School will be an opportunity for young scientists and lecturers from various fields to meet, share their expertise and build a network. For more information, go to https://magnetism.eu/252-2024-school.htm.
Iron Isotopes - Both Radioactive and Stable
July 12, 2023If you ever wondered about doing experiments with different iron isotopes, either in enriched stable form, or with radioactive isotopes, then the product catalog at the National Isotope Development Center (NIDC) could be your source:
https://isotopes.gov/products/Iron?utm_medium=email&utm_source=govdelivery
NIDC can provide enriched Fe-54, Fe-56, Fe-57 and Fe-58, which are all stable, or the radioactive radioisotopes Fe-52 (half-life 8.3 h), Fe-54 (half-life 2.7 years) or Fe-59 (half-life 45 days).

The IEEE Around-the-Clock Around-the-Globe Magnetic Conference
July 05, 2023 The 2023 Around-the-Clock Around-the-Globe Magnetics Conference (AtC-AtG) will be held online on the 27th of September 2023. AtC-AtG is a 24-hour non-stop virtual conference with speakers from all over the world passing the baton to each other, with exciting contributed talks from students and post-doctoral researchers, and intriguing invited talks by emerging and senior scientists. Join, interact, and network at any time – for free.
The 2023 Around-the-Clock Around-the-Globe Magnetics Conference (AtC-AtG) will be held online on the 27th of September 2023. AtC-AtG is a 24-hour non-stop virtual conference with speakers from all over the world passing the baton to each other, with exciting contributed talks from students and post-doctoral researchers, and intriguing invited talks by emerging and senior scientists. Join, interact, and network at any time – for free.
The scope of the conference is to bring together early-stage researchers through a series of oral and poster presentations, coupled with networking opportunities and exciting competitions, providing an international platform to facilitate interaction and discussion of recent progress and trends in the field of magnetism, ranging from fundamental to applied aspects.
The Conference started in 2020 to promote interactions between the younger members of the global magnetism community and has proved to be a huge success. Following that, the 2022 AtC-AtG edition joined together more than 500 scientists, with 50 contributed talks and 65 posters from 25 countries, showcasing both research excellence and diversity.
ERC Starting Grant for Ioana Slabu at RWTH-University Aachen
April 14, 2023
Individuals working in regenerative medicine, cancer treatment, and cardiovascular treatments have great expectations for biodegradable implants. They release active ingredients on schedule, in the right place, at the right time, and in the right dose. Previous attempts failed because no technology is able to properly account for the influence of the aging of the implants in the body. In her project MAD Control, Dr. Ioana Slabu wants to develop a multifunctional platform for hybrid implants with magnetic nanoparticles in dual function: Sensing effect in magnetic imaging and active drug release by magnetic excitation. For the platform, it combines special manufacturing methods with the comprehensive automated characterization of the implants, novel predictive models for their aging, and targeted model-based control of excitation for the first time. To prove the functionality of the platform, a device is being developed that combines the magnetic particle imaging method with a drug-release excitation capability. MAD Control thus promises new opportunities for the research and development of biodegradable implants and magnetic transport and release systems for active ingredients.
Fighting Tumors with Magnetic Bacteria
February 15, 2023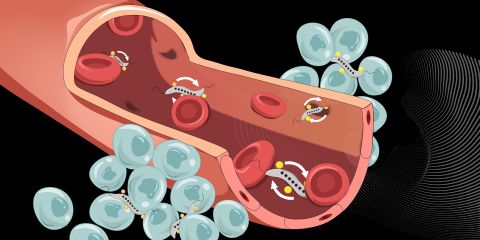 A team of researchers led by Simone Schürle, Professor of Responsive Biomedical Systems at ETH Zurich, have developed a way of controlling certain bacteria to use them as “ferries” to carry anti-cancer drugs through the bloodstream and into tumors. The researchers have chosen to work with naturally magnetic bacteria of the Magnetospirillum genus, which can therefore be controlled from outside the body using magnets. Schürle and her team have now shown that a rotating magnetic field provides three advantages over a static one: its propulsion effect is more powerful; the constant movement it induces in the bacteria makes them more likely to encounter gaps between vessel wall cells; and it does not need to be readjusted once it has been positioned.
A team of researchers led by Simone Schürle, Professor of Responsive Biomedical Systems at ETH Zurich, have developed a way of controlling certain bacteria to use them as “ferries” to carry anti-cancer drugs through the bloodstream and into tumors. The researchers have chosen to work with naturally magnetic bacteria of the Magnetospirillum genus, which can therefore be controlled from outside the body using magnets. Schürle and her team have now shown that a rotating magnetic field provides three advantages over a static one: its propulsion effect is more powerful; the constant movement it induces in the bacteria makes them more likely to encounter gaps between vessel wall cells; and it does not need to be readjusted once it has been positioned.
Gwisai T, Mirkhani N, Christiansen MG, Nguyen TT, Ling V, Schuerle S: Magnetic torque-driven living microrobots for increased tumour infiltration, Science Robotics 26 October 2022, doi: external page10.1126/scirobotics.abo0665call_made
A Telomerase-Activated MRI Probe to Monitor Tumor Growth In Vivo
January 25, 2023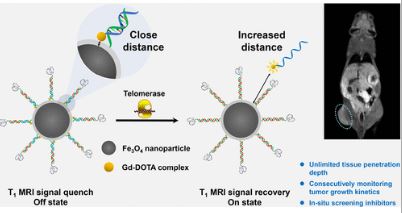
A New Award in the Ferrofluid Community
January 23, 2023
What he needs is a proposal for appropriate award winners. In addition to the name of the nominee and his/her contact information, the proposals should include a maximum one-page justification of the nomination. This justification should outline the particular scientific merits of the nominee as well as his or her contributions to the community at large. Proposals can be submitted in simple text form as a PDF file. For reasons of data security, please refrain from sending text files, WORD files, etc.
When deciding whether to make a suggestion, do not rely on the fact that others will already do it. On the one hand, you never know whether others have the same idea and thus a proposal is omitted in the end, and on the other hand, the information on how often a person has been proposed is also an important indication for the commission of the special merits that is to be honored.
For more information, check out our Archives.
November 2013

Search this site with the power of

